|
10/2/2023 0 Comments Liquid oxygen temperature kelvin 
In the given example we've selected a pressure of 75 atmospheres and a mixture ratio of 2.30, which gives us an adiabatic flame temperature of approximately 3,545 Kelvin. For instance, the curve for a mixture ratio of 2.25 lies approximately midway between the 2.20 and 2.30 curves. If a mixture ratio other than those shown is used, estimate the position of the mixture ratio curve by interpolating between those given. 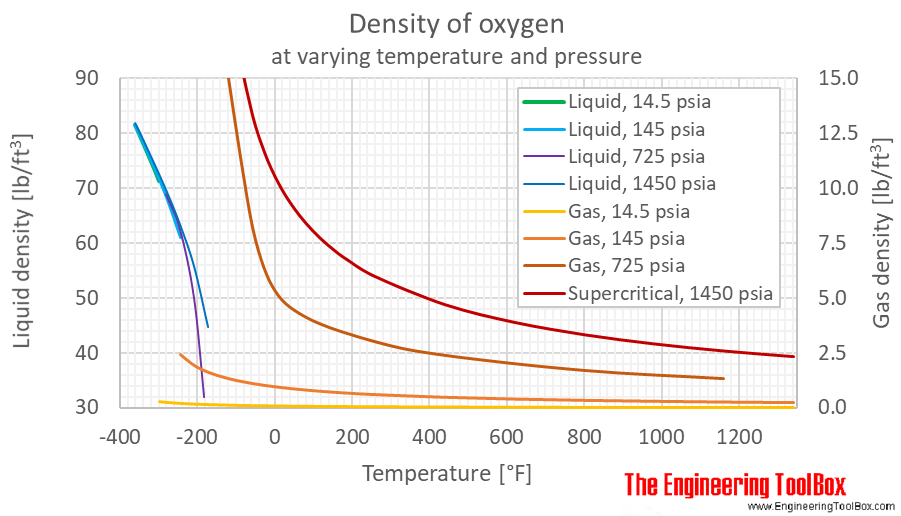
When the vertical line intersects the curve for the desired mixture ratio, draw a horizontal line to the left and read the corresponding adiabatic flame temperature off the vertical axis of the graph. To use this graph, select the desired pressure across the bottom axis of the graph and draw a vertical line. It is the maximum temperature that can be achieved for the given reactants and is used to estimate the combustion chamber temperature (Tc) in a rocket engine.īelow we see a graph of adiabatic flame temperature versus combustion chamber pressure for liquid oxygen and kerosene at three different mixture ratios. In the given example we've selected a combustion chamber pressure of 75 atmospheres and a nozzle exit pressure of 1 atmosphere, which gives us an optimum mixture ratio of 2.30.Īdiabatic flame temperature is the temperature achieved by a combustion reaction that takes place adiabatically, that is, with no heat entering or leaving the system. For instance, the curve for a Pe of 0.7 atmosphere lies approximately one-third the distance from the Pe = 1.0 curve to the Pe = 0.1 curve. If an exit pressure other than those shown is desired, estimate the position of the exit pressure curve by interpolating between those given. When the vertical line intersects the curve for the desired exit pressure, draw a horizontal line to the left and read the corresponding mixture ratio off the vertical axis of the graph. To use this graph, select the desired chamber pressure across the bottom axis of the graph and draw a vertical line. a large section ratio, will have the highest optimum mixture ratio.īelow we see a graph of optimum mixture ratio versus combustion chamber pressure for liquid oxygen and kerosene at two different nozzle exit pressures (Pe). An engine with a high combustion chamber pressure and a low nozzle exit pressure, i.e. A propellant's optimum mixture ratio is a function of the pressures at which the rocket engine will operate. We define the optimum mixture ratio as that which will produce the highest specific impulse for the given reactants. Mixture Ratio is the ratio of oxidizer mass to fuel mass. Adiabatic flame temperature and gas molecular weight have been calculated using the freeware program STANJAN. This data is necessary to determine the velocity of the exhaust gases expelled from a rocket engine, which in turn determines the engine's thrust. These graphs can be used to estimate (1) the optimum mixture ratio of the combustion reactants, (2) the adiabatic flame temperature of the combustion reaction, (3) the average molecular weight of the combustion products, and (4) the specific heat ratio of the combustion products. For each of the propellant combinations shown above, four graphs have been provided.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
